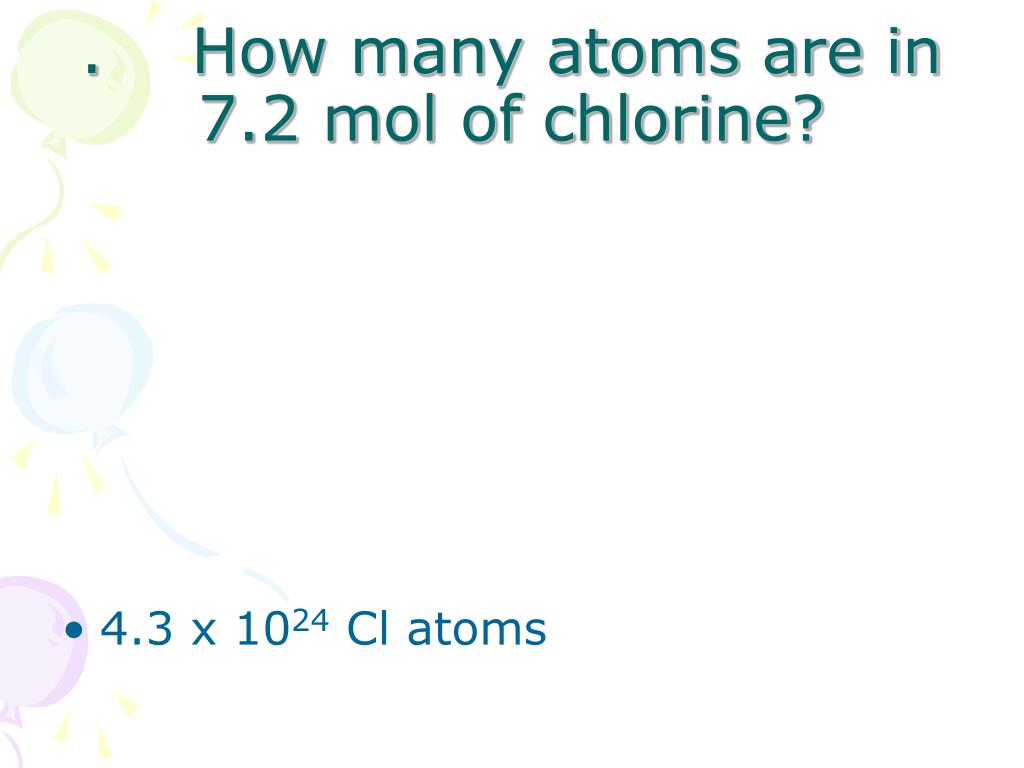
Avogadro’s quantity is describe because the quantity of simple debris (molecules, atoms, compounds, etc.). Although he did now no longer decide the precise percentage, he’s receive credit for the idea.Īvogadro’s quantity is a percentage that relates molar mass on an atomic scale to bodily mass on a human scale. Avogadro’s NumberĪmadeo Avogadro first propose that the extent of a fonline at a given strain and temperature is proportional to the quantity of atoms or molecules. This comes out to 58.44276 grams according to mole which we will spherical to 58.forty four grams according to mole. Then, we upload those hundreds collectively to locate the overall mass of sodium chloride molecules. Next, we do the equal for the mass of chlorine atoms, that’s 35.453 grams according to mole. Now that we recognize what number of atoms there are for every detail, we will locate the molar mass.įirst, we calculate the mass of the sodium atoms, that’s 22.98976 grams according to mole. Step 2: Count what number of atoms there are for every detailįor the compound sodium chloride, considering there aren’t anyt any subscripts (small numbers at the lowest of every detail’s image), meaning there’s simplest one sodium and simplest one chlorine atom for this compound. The detail chlorine has an atomic mass of 35.453 g/mol. The detail sodium has an atomic mass of 22.98976 g/mol. If you have a take a observe the entries for sodium and chlorine within side the table, we can take note of the numbers that seem within side the pinnacle left (see video). 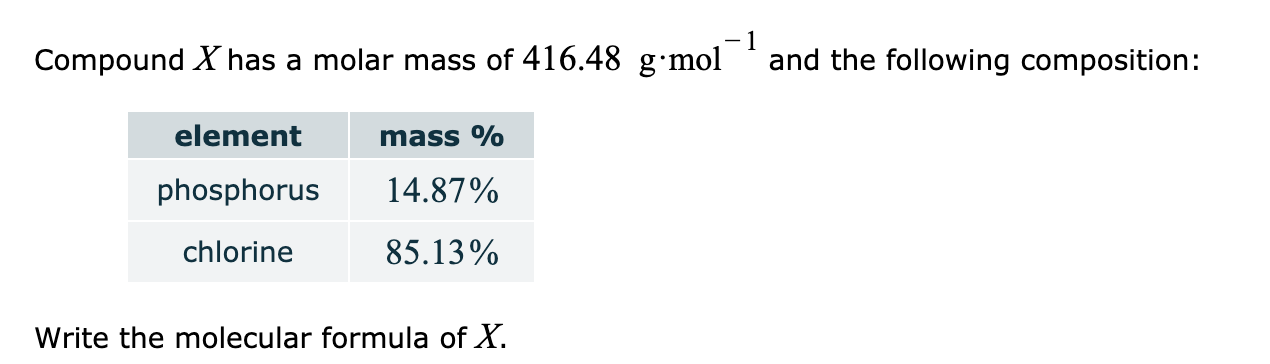
The first issue we want is to locate the person atomic hundreds for every detail. Step 1: Find the atomic hundreds of person factors withinside the periodic table The first issue we want to do is to locate sodium and chlorine within side the periodic table. Let’s use this compound, sodium chloride, as our first instance on a way to calculate the molar mass for the compounds. The compound sodium chloride is made of factors, namely, sodium and chlorine. For instance, a few not unusual place compounds consist of salt, glucose, acetic acid (or vinegar), and sodium bicarbonate (or baking soda). How to Find the Molar Mass for CompoundsĬompounds are materials which might be made of multiple detail. The molar mass, additionally recognized as molecular weight, is the sum of the overall mass in grams of all of the atoms that make up a mole of a specific molecule. The unit used to degree is grams according to mole. If we already recognize the quantity of moles needed, we will use the idea of molar mass to calculate what number of grams of the substance are required. A mole is a unit of degree that enables us evaluate debris of any given substance and its mass. Through cautious calculation, scientists can decide the quantity of moles a selected response will want. But, how are we able to degree some thing so small in an correct way? How will we commonly degree molecules? In the technology laboratory, we use a device known as an analytical balance to degree in grams. Substances take in area and feature mass. Molecules, which make up materials, regularly want to be measured in experiments, and it’s miles critical that those measurements are correct. Step 2: Count what number of atoms there are for every detail.Step 1: Find the atomic hundreds of person factors withinside the periodic table.How to Find the Molar Mass for Compounds. 
And don’t forget to put the unit g/mol to your final calculated molar mass.First solve the brackets, then multiplications and at last do the final addition. Always follow the calculation order to avoid any mistakes in calculation. 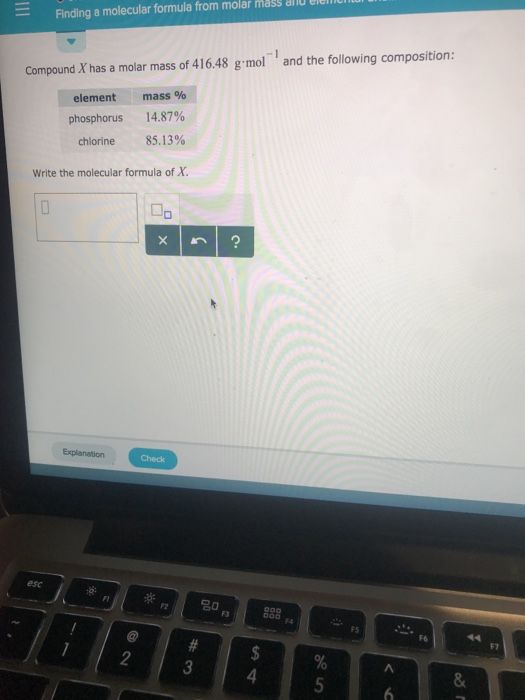
But all these units (i.e g/mol, grams/mole and g/mole) are the same.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
